Science & Technology - 2016
May
Solving the Challenges of Making Liquid-Hydrogen Targets
Just like water, hydrogen can exist in three different states, or phases—solid, liquid, and gas—depending on the temperature and pressure. Both solid and liquid phases can be used in NIF inertial confinement fusion (ICF) and high energy density (HED) implosions, and each has its own distictive advantages.
Most NIF ICF experiments to date have relied on the formation of a thin solid layer of the hydrogen isotopes deuterium and tritium on the inside surface of the target capsule at a temperature of 18 to 20 kelvin (-427 to -424 degrees Fahrenheit). Cryogenically cooling the DT fuel to just below the freezing point of hydrogen, which changes hydrogen’s phase from a gas to a liquid and finally to a solid, enhances the density of the fuel and improves the chances of achieving the required density for ignition.
But growing acceptable DT ice layers can be difficult and time-consuming, and the rocket-like implosion of the solid fuel results in a high convergence ratio (the initial fuel radius divided by the imploded radius). Researchers believe this could contribute to fuel instabilities and asymmetries that can interfere with a successful fusion “burn” in the hot spot at the center of the capsule. On the other hand, a lower convergence ratio also produces a lower energy yield.
In an effort to develop and test a liquid-layer approach as an additional experimental platform for NIF, members of LLNL’s Materials Science Division and target specialists at General Atomics have spent several years painstakingly developing a technique to line NIF target capsules with a uniform polymer foam that can be saturated with liquid DT. The “wetted-foam” targets were put to their first test on April 21-22, with good results, by a team led by researchers from Los Alamos National Laboratory (see “NIF Liquid-Hydrogen Target Gets Its First Test”)
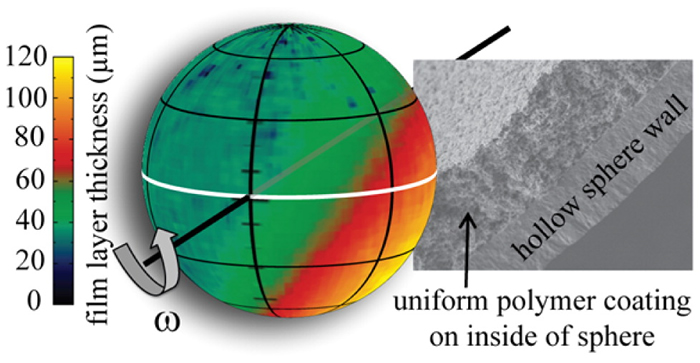 In order to fabricate wetted-foam targets, LLNL materials scientists used image analysis and computer fluid dynamics simulations to explore the effect of liquid viscosity and rotational velocity on film uniformity. The data were then used to demonstrate the fabrication of uniform sol-gel chemistry-derived porous polymer films inside two-millimeter inner-diameter diamond shells.
In order to fabricate wetted-foam targets, LLNL materials scientists used image analysis and computer fluid dynamics simulations to explore the effect of liquid viscosity and rotational velocity on film uniformity. The data were then used to demonstrate the fabrication of uniform sol-gel chemistry-derived porous polymer films inside two-millimeter inner-diameter diamond shells. Researchers say replacing the ice layer with liquid hydrogen could provide several benefits: a lower convergence ratio; increased flexibility in creating the initial conditions of the implosion, in order to gain insights into hot-spot formation; the ability to bring dopants (small, controlled amounts of impurities) into direct contact with the DT fuel for diagnostic and nuclear physics purposes; and potential increases in NIF’s shot rate by avoiding the time-consuming DT ice layering process.
Liquid hydrogen, however, has its own issues—primarily instabilities caused by the liquid “slumping” during the implosion from the effects of gravity. Nearly 30 years ago, LLNL researchers Dale Darling and Richard Sacks proposed a solution: line the inside of the target capsule with a low-density foam that would act like a sponge, holding and stabilizing the liquid hydrogen against gravitational slumping.
While foam-lined capsules were subsequently tested in direct-drive implosion experiments, in which the target capsule is directly irradiated by laser beams, target fabricators faced a number of challenges in adapting the technology for NIF’s indirect-drive experiments using capsules suspended inside hohlraums. One goal of the wetted-foam method—bringing dopants into direct contact with the fuel—required thinner and lower-density foam shells in combination with a much thicker ablator shell than those previously used.
Rather than trying to create free-standing foam shells, the researchers explored a new approach: using prefabricated high-density carbon, or diamond, ablator shells as molds to cast concentric thin-walled low-density foam shells using sol-gel chemistry. In the sol-gel process, simple molecular precursors are converted into nanometer-sized particles to form a colloidal suspension, or sol. The molecular precursor solution is inserted into the capsule through a tiny fill tube or a pressure filling process, where it transforms into a high-viscosity polymer fluid and then gels, creating the foam.
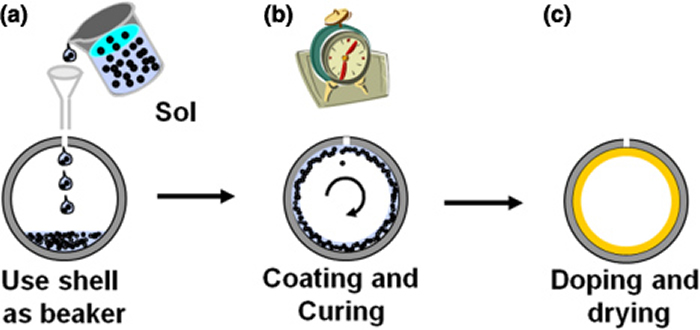 Schematic of the fabrication process for indirect-drive foam-lined ICF targets: (a) filling of a prefabricated ablator shell with the desired amount of an aerogel precursor solution, (b) formation of a smooth and uniform gel layer by deterministic rotation of the capsule during polymerization, and (c) doping and supercritical drying.
Schematic of the fabrication process for indirect-drive foam-lined ICF targets: (a) filling of a prefabricated ablator shell with the desired amount of an aerogel precursor solution, (b) formation of a smooth and uniform gel layer by deterministic rotation of the capsule during polymerization, and (c) doping and supercritical drying. “There were issues everywhere,” said LLNL materials scientist Juergen Biener. “The sol-gel process is performed in liquid phase, and thus one needs to fill the premade ablator shells with a precise amount of precursor solution. These are very, very tiny amounts, much less than a normal water droplet. Inventing a method to fill these small amounts reproducibly and deterministically into the shell was one of the challenges we worked on.”
Part of the problem was the need for a low-density gel chemistry that could tolerate the motion of the capsule as it rotated to allow the precursor solution to form a uniform liquid layer on the inside surface of the shell. “The material has to flow around while it’s gelling,” Biener said. “If we used the normal, standard gel chemistry, that gel would not survive. So we had to develop a gel chemistry for these very low-density gels that tolerated mechanical motion during gelation.”
Finding the best way to rotate the capsule was another challenge, added LLNL scientist Tom Braun. “Once we tuned the chemistry to the required specifications,” he said, “we had to find the corresponding rotation parameters for the shell that contains the liquid precursor solution; you have to rotate it in a specific way to form a uniform foam layer on the inside.
“In the beginning we rotated the shell around two axes at the same time, but we learned with the help of simulations that uniaxial rotation can result in the formation of very uniform thin film layers in hollow spherical shells. It’s almost counterintuitive if you think about it, but the experiments confirmed it. That was a big discovery for us, a big step forward.”
 LLNL materials scientist Tom Braun fills a diamond capsule with aerogel precursor solution using a self-developed pressure filling setup. A small vacuum chamber contains a vial with the precursor solution and a linear feedthrough to which the ablator shell is attached (see inset). To achieve high reproducibility with high accuracy, the chamber is under-pressurized while the capsule is not in contact with the precursor solution. The applied pressure differential determines the foam layer thickness. The capsule is then submerged in the precursor solution and filled by re-pressurizing the chamber to atmospheric pressure. This pressure differential filling setup has the required accuracy to precisely control the foam layer thickness in a spherical shell and can easily be modified for different shell sizes and volumes. Credit: James Pryatel
LLNL materials scientist Tom Braun fills a diamond capsule with aerogel precursor solution using a self-developed pressure filling setup. A small vacuum chamber contains a vial with the precursor solution and a linear feedthrough to which the ablator shell is attached (see inset). To achieve high reproducibility with high accuracy, the chamber is under-pressurized while the capsule is not in contact with the precursor solution. The applied pressure differential determines the foam layer thickness. The capsule is then submerged in the precursor solution and filled by re-pressurizing the chamber to atmospheric pressure. This pressure differential filling setup has the required accuracy to precisely control the foam layer thickness in a spherical shell and can easily be modified for different shell sizes and volumes. Credit: James Pryatel Still another issue was making sure the foam would tolerate being wetted by the liquid hydrogen, which is cryogenically cooled to temperatures in the range of 21 to 26 kelvin (-421 to -413 degrees Fahrenheit). Braun and LLNL colleagues Bernie Kozioziemski and Tony van Buuren conducted numerous experiments to see how different gel chemistries responded to wetting. One early candidate, an extremely low density silica aerogel, could not tolerate the wetting and collapsed. “But the polymer aerogel which we developed was very robust against wetting,” Biener said.
The liquid DT presented yet another challenge as a layered target platform. “The liquid can move more easily in and out of the capsule compared to a solid DT layer,” Kozioziemski said. “The wetted foam target was designed with a thermal switch that allows liquid to flow in during filling with the switch open, and then freezes a solid DT plug in the fill-line when the correct amount of liquid is in the shell.”
The foam-lined capsules are fabricated at LLNL, then shipped to General Atomics for fill-tube attachment and cleaning. The final targets are assembled in the Laboratory’s target fabrication facility. The researchers said future work will focus on further improving the uniformity of the foam shells, increasing the yield of the process, and exploring alternative aerogel chemistries.
Along with Biener, Braun, and Kozioziemski, team members who contributed to the foam-lined capsule development include LLNL’s Christoph Dawedeit, Sung-Ho Kim, Marcus Worsley, Alex Chernov, Christopher Walton, Trevor Willey, Sergei Kucheyev, Swanee Shin, Morris Wang, Monika Biener, Jon Lee, Tony van Buuren, Kuang Jen Wu, Joe Satcher, and Alex Hamza; and Neal Rice, Casey Kong, Jay Crippen, and Haibo Huang of General Atomics.



